Poliovirus surveillance in Ghana
Project Lead(s)

Associate Professor
Project Background
Poliomyelitis, or polio for short, is a highly infectious viral disease. The virus, which mainly affects children under five, attacks the brain and spinal cord and can cause total paralysis within days of infection. There are three poliovirus serotypes, Type 1, 2 and 3 all of which have identical symptoms. The virus transmission is mainly through the faecal-oral route, or food or water that is contaminated with the virus. Environmental factors including poor sanitation can also help spread the virus.
The Ghana Polio Regional Reference Laboratory supports the Global Poliomyelitis Eradication Initiative by the vital role it plays in the detection and monitoring circulation of wild and vaccine-derived poliovirus in Ghana and the West Africa sub-region. Stool from acute flaccid paralysis (AFP) cases and sewage/wastewater from environment sites (ES) are the main samples investigated in the laboratory
Objectives/Research Areas
- To provide virus isolation, intratypic differentiation and sequencing of polioviruses from acute flaccid paralysis (AFP) and environmental surveillance.
- To conduct sero-epidemiological survey and sharing of data with National Program on Immunization, WHO Country Office and WHO/AFRO.
Approach:
Once a case of AFP has been reported, two stool samples are collected from the AFP patient, 24 hours and 48 hours apart, ideally within 14 days of the onset of paralysis, and transported to the nearest dedicated laboratory. Results must be reported within 14 days, allowing countries to take action immediately to limit virus spread. For environment surveillance, sewage or wastewater sample collection is scheduled based on epidemiological week and frequency is one collection every four weeks per site. Results must be reported within 21 days.
Key Findings
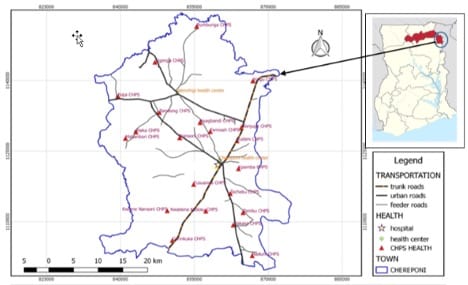
The Polio lab processed AFP samples from three countries namely, Ghana, Benin, Togo from 2019 to 2020. In 2021, the lab further received samples from three more countries, Liberia, Sierra Leone and Niger to make the total number of countries six. A total of 10,868 AFP and 1445 environmental samples were received and processed. The non-poliovirus isolation rate was 13.3% for the 3-year period. The country experienced an outbreak of circulating vaccine-derived poliovirus type 2 (cVDPV2) from 2019 to 2020. The first cVDPV2 was detected from environmental wastewater (figure 1) in the Northern region. This was followed by the first human case from Chereponi District in the Savanna region (figure 2). A total of 19 cVDPV2 from AFP cases was confirmed from Ahafo, Ashanti, Bono, Northern, Oti, and Savanna regions in 2019. cVDPV2 was also confirmed from 21 of their contacts in 2019. In 2020 however, the lab confirmed 12 cVDPV2 from AFP cases and 10 from their contacts in 5 regions. From environmental surveillance, 17 and 18 cVDPV2 were confirmed respectively in 2019 and 2020.

The country responded to the outbreak by conducting vaccination campaigns with the monovalent oral polio vaccine (mOPV2) in 2019 and 2020 and transmission was interrupted.
The polio lab also isolated and confirmed through genomic sequencing cVDPV2 for Benin and Togo in 2019 and 2020 from both AFP and environmental surveillance. In 2021 the polio lab isolated 34 nOPV2 from AFP and 32 from environmental samples received from Sierra Leone, Benin and Liberia who are using the novel oral polio vaccine to respond to the cVDPV2 outbreak.
Ongoing Activities
Virtual Online Training with CDC team (12th November – 19th November 2021)
Detection of poliovirus directly from stool specimens is a new, faster methodology for poliovirus identification that will help reduce laboratory analysis time by up to one week.
In line with the GPEI’s objective on Polio Surveillance 2022-2026 to improve detection and response through sensitive surveillance, the polio lab participated in a training on Direct detection of polio viruses organized (figure 3) by CDC virtually for Ghana and other polio labs in the GPLN. The first was on 12th to 19th November 2021, followed by 1st February 2022 and 3rd February 2022. Six Polio lab staff participated in these trainings.
Interns and National Service Personnel
A total of 20 interns from universities across the country as well as other nationals were trained over the period. Furthermore, 15 National Service Personnel were also trained.
Technical Meetings: The RRL participated in the National Polio Expert Committee, National Certification Committee and the National Task Force on Polio. The head of the polio lab also participated on the National Technical Coordinating Committee’s meetings.
Weekly Report: Weekly laboratory reports were sent to EPI, Surveillance, WHO country office and WHO HQ to up-date all partners for programmatic considerations.
Data Cleaning and Reconciliation: The RRL is part of the national polio team that met regularly for data cleaning and data reconciliation within the period under review.

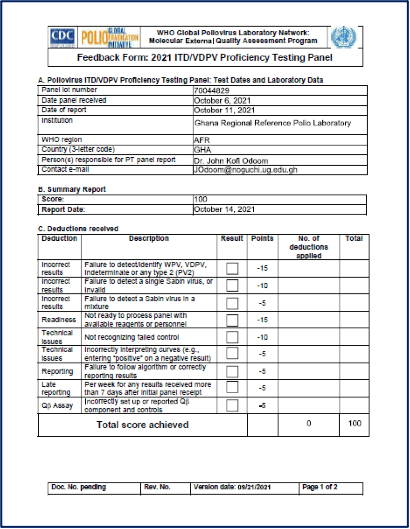
Accreditation: The last time the Polio laboratory had on-site WHO annual accreditation visit was in 2017. The Polio lab has however participated in all proficiency testing panes to date. The lab scored 100% (figure 4) in 2021 intratypic differentiation proficiency panels. We are waiting for the isolation panels on AFP and ES and onsite accreditation in June 2022.
Key Publications
- Odoom JK, Obodai E, Boateng G, Diamenu S, Attiku K, Avevor P, Duker E, Boahenea B, Eshun M, Gberbi E, and Opare JKL. Detection of vaccine-derived poliovirus circulation by environmental surveillance in the absence of clinical cases. Hum Vaccin Immunother 2021 Feb. https://doi.org/10.1080/21645515.2020.1852009
- Opare JKL, Odoom JK, Akweongo P, Afari EA, Pappoe M Poliovirus antibody levels and lameness among individuals in three regions of Ghana. Hum Vaccin Immunother. 2019 Jul 2. doi: 10.1080/21645515.2019.1637235
- Opare JKL, Akweongo P, Afari EA, Odoom JK. Poliovirus neutralizing antibody levels among individuals in three regions of Ghana. GMJ. 2019, 53(2);170-180. Doi:http://dx.org/10.4314/gmj. v53i2.13
Community Engagement Activities
1.Th Polio Lab has been conducting stool survey in districts and communities that are silent for submitting AFP samples
2.The Polio Lab also partner the Disease Surveillance Department and Expanded Programme on Immunization to conduct outbreak investigations in communities when outbreaks occur.
Team
External Collaborator(s)
Ghana Health Service (GHS)
National Public Health and Reference Laboratory (NPHRL)
Expanded Programme on Immunisation, Centre for Disease Control and Prevention (CDC)
National Institute for Biological and Standards Control (NIBSC)
World Health Organization (WHO)
Funder(s)
World Health Organisation
CDC